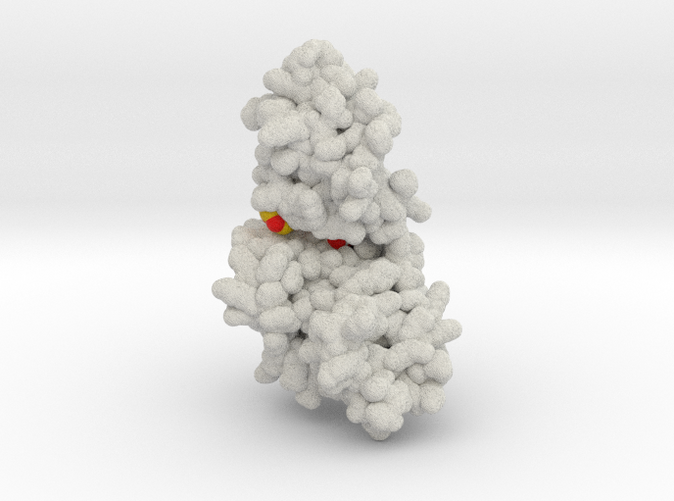
Model Description
This is a 3D printed protein model of DYRK2 (dual-specificity tyrosine-regulated kinase 2) bound by Curcumin. This model visualizes the binding properties of Curcumin (yellow-heteroatom type) for DYRK2's (white) by the identifying the three binding residues deep inside the ATP binding pocket, Asp368, Glu266, Lys251. Each residue is colored by heteroatom type. Model created from PDB ID: 5ZTN and designed to pair with original PNAS publication:
https://www.pnas.org/content/early/2018/07/05/1806797115
Protein Description
Ancient drug curcumin impedes 26S proteasome activity by direct inhibition of dual-specificity tyrosine-regulated kinase 2
Curcumin is an ancient drug derived from turmeric and has been found to exhibit potent anticancer properties albeit through controversial mechanisms of action. Using a biochemical model, mouse cancer model, and cellular models, we show that curcumin is a highly potent and selective inhibitor of dual-specificity tyrosine-regulated kinase 2 (DYRK2), a positive regulator of the 26S proteasome. Curcumin perturbs 26S proteasome activity via DYRK2 inhibition in various cancer cells and in the mouse cancer model leading to impairment of cell proliferation and reduction of cancer burden in mice. This novel mechanism of action of curcumin opens up new avenues for potential preventative or therapeutic strategies in proteasome-addicted cancers like triple-negative breast cancer and multiple myeloma.
Structure of DYRK2 in Complex with Curcumin.
To elucidate how curcumin specifically inhibits DYRK2, we crystallized DYRK2 in the presence of curcumin and determined the structure at 2.5 Å (
SI Appendix, Table S2) [Protein Data Bank (PDB) ID code: 5ZTN]. Strong electron densities are present for curcumin, allowing confident interpretation of its position (
Fig. 2A). Curcumin occupies the ATP-binding pocket of DYRK2 (
Fig. 2B). One of the 4-hydroxy-3-methoxyphenyl groups of curcumin forms hydrogen bonds with the Lys251 (the ion pair Lys), Glu266 (the ion pair Glu), and Asp368 (the DFG Asp) of DYRK2 that anchors curcumin deep within the ATP-binding pocket of DYRK2 (
Fig. 2C). Ile228, Ala249, Ile285, Phe301, Leu303, Leu355, and Ile367 are involved in making hydrophobic interactions with curcumin (
Fig. 2C). The cocrystal structure of DYRK2-curcumin clearly reveals that curcumin imparts its inhibitory effect on DYRK2 by directly binding to the DYRK2 ATP-binding pocket.
https://www.pnas.org/content/early/2018/07/05/1806797115